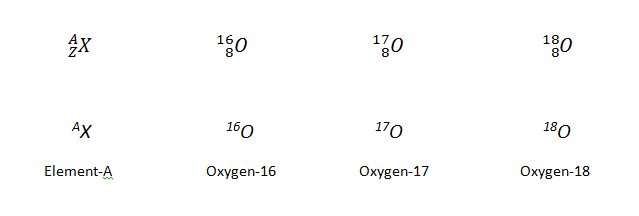Tags | |
UUID | 1a0a3443-f145-11e9-8682-bc764e2038f2 |
Introduction
From UCDavis Chemwiki
Nuclear reactions differ from other chemical processes in one critical way: in a nuclear reaction, the identities of the elements change. In addition, nuclear reactions are often accompanied by the release of enormous amounts of energy, as much as a billion times more than the energy released by chemical reactions. Moreover, the yields and rates of a nuclear reaction are unaffected by changes in temperature, pressure, or the presence of a catalyst.
The Atomic NucleusAs you learned in General Chemistry I, each element can be represented by the notation AZX, where A, the mass number, is the sum of the number of protons and the number of neutrons, and Z, the atomic number, is the number of protons. The protons and neutrons that make up the nucleus of an atom are called nucleons, and an atom with a particular number of protons and neutrons is called a nuclide. Nuclides with the same number of protons but different numbers of neutrons are called isotopes. Isotopes can also be represented by an alternative notation that uses the name of the element followed by the mass number, such as carbon-12. The stable isotopes of oxygen, for example, can be represented in any of the following ways:
Because the number of neutrons is equal to A ? Z, we see that the first isotope of oxygen has 8 neutrons, the second isotope 9 neutrons, and the third isotope 10 neutrons. Isotopes of all naturally occurring elements on Earth are present in nearly fixed proportions, with each proportion constituting an isotope’s natural abundance. For example, in a typical terrestrial sample of oxygen, 99.76% of the O atoms is oxygen-16, 0.20% is oxygen-18, and 0.04% is oxygen-17.Any nucleus that is unstable and decays spontaneously is said to be radioactive, emitting subatomic particles and electromagnetic radiation. The emissions are collectively calledradioactivity and can be measured. Isotopes that emit radiation are called radioisotopes. |
This Collection is empty
- Comments
- Attachments
- Stats
No comments |