The Half-Life of Zero Order Reaction calculator computes the half-life in nuclear decay for a zero order reaction.
INSTRUCTIONS: Enter the following:
- (k) This is the temperature-dependent reaction rate constant.
- (A0) This is the initial concentration
Half-Life: The calculator returns the half-life.
Chemistry Rate Law Calculators
- Zero Order Rate Law (Integral form)
- Zero Order Half Life
- Zero Order Rate Law
- First Order Rate Law (Integral form)
- First Order Half Life
- First Order Rate Law
- Second Order Rate Law (Integral form)
- Second Order Half Life
- Second Order Rate Law
The Math / Science
The half-life of a chemical reaction is defined as the time required for half the amount of a reactant to be converted into product. To find the half-lives of different order reactions, we use integrated rate laws and rate constants to relate concentration to time. There are three different rate laws that can be used to find the half-life of a chemical reaction: zero, first, and second order.
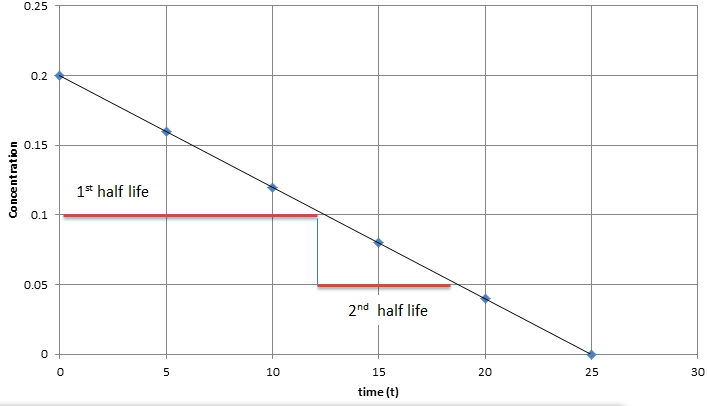
For zero order kinetics, the half-life is not dependent on the concentration of the substance. In other words, rate = k. This means that no matter how much substrate is added, and no matter how high of a concentration is present in solution, the rate of the reaction will not increase. A graphical representation of this would show linear data with a negative slope, as shown in the image below. The rate law for a zero order reaction is [A] = [A]0 - kt. To find the half-life for a zero order reaction, the equation t1/2 = [A]0 / 2k is used.
Where
- k is the temperature-dependent reaction rate constant
- t1/2 is the half-life
- [A]0 is the initial concentration
References
Whitten, et al. "Chemistry" 10th Edition. Pp. 631